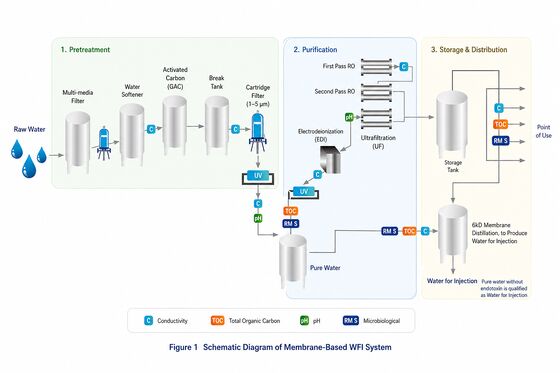
Membrane-Based WFI System
Szczegóły Produktu:
| Miejsce pochodzenia: | Chiny |
| Nazwa handlowa: | CHONGYANG |
| Orzecznictwo: | ISO ,CE |
| Numer modelu: | CY-WFI-300 litrów/godz |
Zapłata:
| Minimalne zamówienie: | 1 |
|---|---|
| Cena: | negotiable |
| Szczegóły pakowania: | Zgodnie ze standardem eksportowym |
| Czas dostawy: | Z 30-40 dniami |
| Zasady płatności: | Akredytywa, T/T |
| Możliwość Supply: | 100 zestawów/miesięcy |
|
Szczegóły informacji |
|||
| Tworzywo: | SS316L | Gradacja: | Podwójna ultrafiltracja RO +EDI+ |
|---|---|---|---|
| Zawór: | Zawór membranowy | Pompa: | Grundfos, CNP |
| Przewodność: | 1,3 μS/cm | Wymagana woda zasilająca: | System wody oczyszczonej |
| Nazwa produktu: | Ultrafiltracja wody WFI | Standard: | FDA, USP, cGMP, GMP |
| Materiał rurociągu: | BPE, SS | Pojemność: | 100L/H -10 000L/H |
opis produktu
Membrane-Based WFI System
Product Overview: The Membrane-Based WFI System is designed for pharma Water for Injection production using double-pass RO, EDI and ultrafiltration technology. It provides stable water quality, lower energy consumption and GMP-compliant operation for modern pharmaceutical manufacturing.
Applications
This system is widely used in pharma manufacturing, biotechnology production, sterile preparation, laboratory research and medical industries where high-purity Water for Injection is required for injection, cleaning and process water applications.
Process Flow
Technical Parameters
| Item | Specification |
|---|---|
| Capacity | 100–10,000 L/H |
| Conductivity | ≤1.1 µS/cm @25°C |
| TOC | ≤500 ppb |
| Endotoxin | ≤0.25 EU/ml |
| Bacteria | <10 CFU/100 ml |
| Material | SS316L, internal Ra <0.4 μm |
| Control System | PLC + HMI, or Schneider optional |
Configuration List
The system includes multimedia filter, activated carbon filter, water softener, double-pass RO membranes, EDI module, ultrafiltration membrane for endotoxin removal, sanitary pumps, UV sterilizer, ozone system, SS316L storage tank and automatic PLC control system.
Project Cases
Our membrane-based WFI systems have been applied in pharmaceutical factories in Southeast Asia, the Middle East and Europe, helping customers achieve stable WFI production, GMP validation and continuous operation.
Certifications & Standards
The system is designed according to GMP, FDA, USP, EP and WHO requirements. Optional IQ/OQ/PQ validation support and complete documentation can be provided for audit and compliance needs.
FAQ
Q: Is membrane-based WFI acceptable under GMP?
A: Yes. It can be accepted with proper system design, validation and water quality control strategy.
Q: What are the advantages compared with distillation?
A: Lower energy consumption, compact design, faster startup and flexible operation.
Q: Can the system be customized?
A: Yes. Capacity, materials, control system and validation documents can be customized.
![]()
![]()
FAQ:
1.Question: Which process do you adopt ?
Answer:Usually we will be according to what your raw water quality and your requested water requirement .
2. Question:Can you customize For me?
Answer :Yes. We can manufacture according your requirement , we can even design appearance of equipment .
3.Question: What standards is the Pharmaceutical Purified Water System ?
Answer: Usually there are five standards in this pharmaceutical industry , FDA,cGMP ,GMP ,USP, Japanese .
4.Question:How long is the Delivery time ?
Answer: Usually the delivery time is 35 -40 Days .